How to Ensure Surgical Implant Quality Control for Optimal Patient Safety?
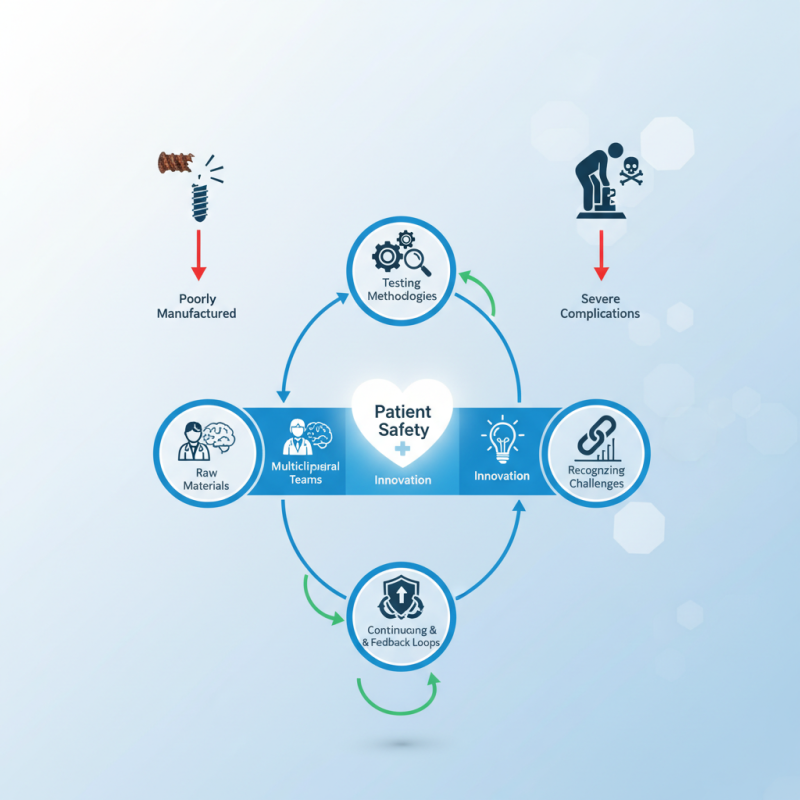
In the realm of modern medicine, surgical implant quality control is paramount. Ensuring the integrity of implants directly impacts patient safety. Poorly manufactured implants can lead to severe complications. Thus, comprehensive quality control measures are essential.
Various factors influence surgical implant quality control. Raw materials must meet stringent standards. Testing methodologies need to be rigorous. However, even with proper processes, lapses can occur. Continuous monitoring and feedback loops are crucial for improvement. Engaging multidisciplinary teams can enhance oversight.
Healthcare professionals must remain vigilant. Patient safety should always be the priority. The industry must embrace innovation to improve quality control. Yet, complacency may creak in, and past successes can foster a false sense of security. Recognizing these challenges is the first step toward better outcomes.
Understanding the Importance of Surgical Implant Quality Control
Surgical implant quality control is vital for patient safety. It ensures that implants are not only effective but also safe for use. Each implant must meet rigorous standards. This prevents complications and enhances the overall success rate of procedures. High-quality implants lead to better recovery outcomes and improve patients' quality of life.
However, maintaining these standards is challenging. Flaws can occur during manufacturing or even during storage. Continuous monitoring and testing are essential. Each batch of implants should be evaluated thoroughly. Training staff to recognize potential issues is equally important. This way, they can address problems before they reach patients.
Patients place immense trust in their healthcare providers. They expect safe and reliable implants. Transparency in quality control processes builds this trust. Sharing information about inspections and standards used can foster confidence. Addressing imperfections in the process opens up discussions for improvement. Continual reflection on these practices leads to better patient care. Quality control is not merely a requirement; it is a commitment to patient safety.
Key Standards and Regulations Governing Surgical Implants
Ensuring quality control in surgical implants is crucial for patient safety. Several key standards and regulations guide the manufacturing and testing of these medical devices. The International Organization for Standardization (ISO) plays a significant role by defining specific criteria for implant safety and effectiveness. Compliance with ISO standards can help minimize risks associated with infections or device failures.
Reviewing the FDA guidelines is essential for manufacturers. The FDA demands thorough testing, including biocompatibility and mechanical integrity assessments. Adhering to these regulations helps ensure that implants are safe for use. Skilled teams must create comprehensive documentation to demonstrate compliance with these standards.
**Tip:** Regular audits of manufacturing processes can reveal potential issues. This proactive approach allows teams to address problems before they compromise safety.
Implementing strict quality control measures is vital. However, not all manufacturers may meet these standards consistently. It's important for healthcare professionals to verify that implants meet regulatory requirements.
**Tip:** Always request third-party certifications. Independent verification can enhance credibility and ensure that patient safety remains a priority.
Critical Steps in the Quality Control Process for Surgical Implants
Surgical implant quality control is critical to ensure patient safety. The quality control process begins with rigorous material selection. According to a 2022 report by the World Health Organization, 70% of implant failures are linked to material defects. This highlights the need for precise testing methods.
Next, manufacturing processes must adhere to strict guidelines. The ISO 13485 standard requires comprehensive documentation and traceability. This ensures that every step of the production is tracked. However, some manufacturers still struggle with compliance. Roughly 30% miss key quality checks, leading to potential risks.
Finally, post-market surveillance is essential. Regular audits and feedback loops can catch issues early. Data from the FDA shows that 15% of reported adverse events stem from design flaws. Therefore, implementing a robust feedback mechanism is crucial for ongoing quality assurance. Aiming for continuous improvement can help address existing gaps and enhance patient safety effectively.
Approaches for Testing and Assessing Implant Biocompatibility
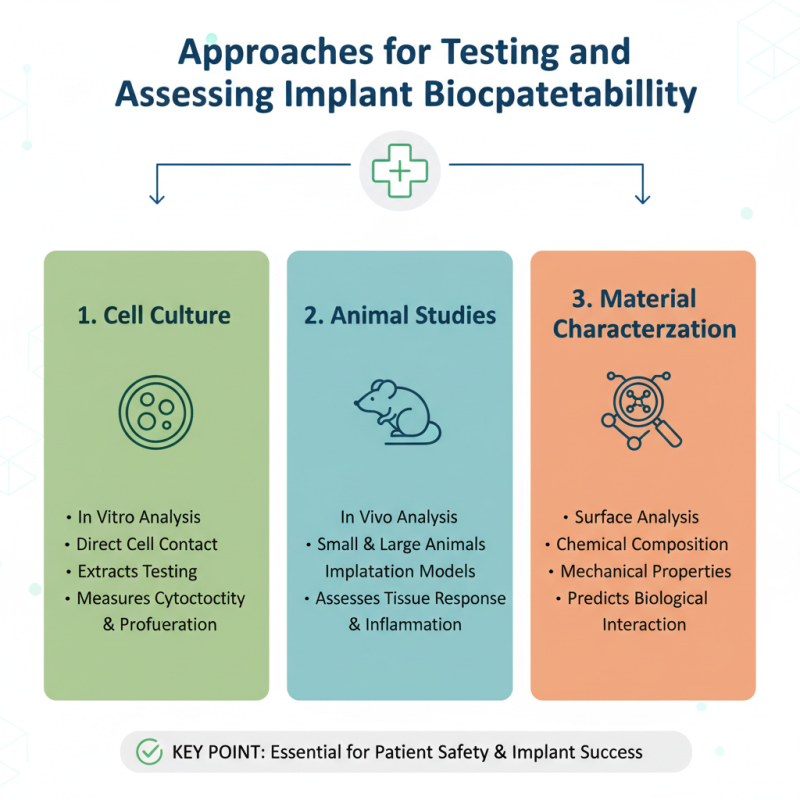
Ensuring implant biocompatibility is critical for patient safety. Testing materials before use is essential. Various methods assess how implants interact with biological systems. Standard tests include cell culture, animal studies, and in vitro analyses. Each approach has strengths and weaknesses that require careful consideration.
Cell culture studies provide a controlled environment to observe cell responses to materials. However, this method may not fully replicate the complexities of the human body. Animal studies offer more insight but can introduce ethical dilemmas and variability. Additionally, transitioning results from animals to human applications remains challenging.
Selecting the right biocompatibility tests can be complex. Not all materials behave consistently across different tests. Regulatory guidelines often evolve, making it difficult to keep up. The need for continual reassessment of testing methods is crucial. Researchers must strive to improve protocols, ensuring the highest quality standards are met. Attention to these details directly impacts patient outcomes.
Continuous Monitoring and Feedback for Improved Patient Outcomes
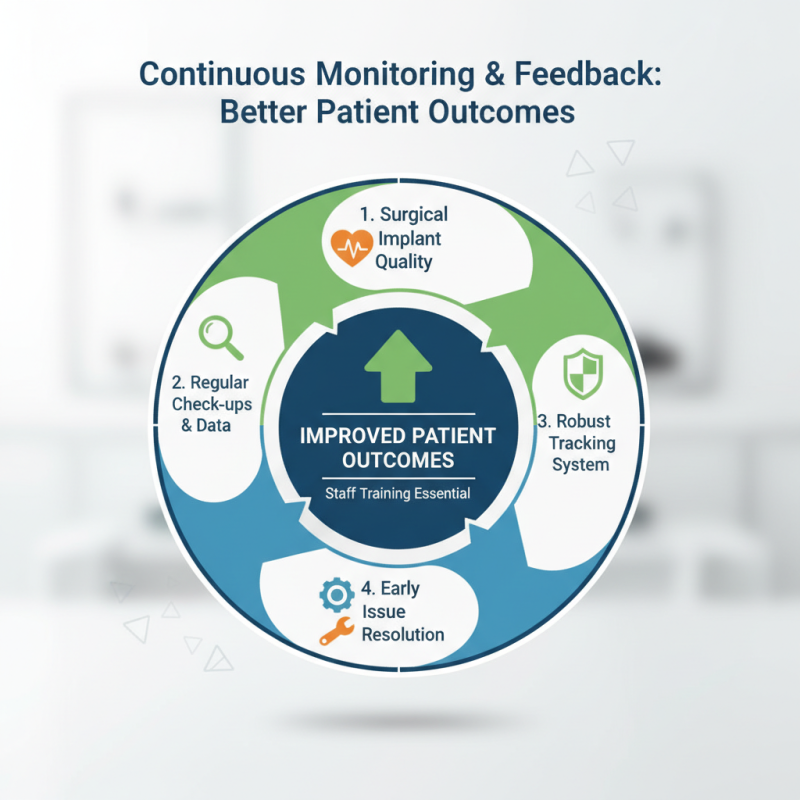
Continuous monitoring is crucial for enhancing surgical implant quality. Regular check-ups and data collection help identify potential issues early. Hospitals must employ robust tracking systems for implants. This ensures that any abnormalities are quickly addressed. Staff training is essential for effective monitoring and feedback.
Feedback loops between healthcare providers and manufacturers foster accountability. This collaboration can lead to constant improvements in implant design and safety. Real-time data can highlight trends and indicate necessary modifications. However, inconsistencies in reporting can undermine this valuable process. Encouraging open communication is paramount.
Reflecting on adverse events offers lessons for future practices. It is vital to create an environment where staff can share concerns without fear. Continuous education on established protocols can boost confidence in implant procedures. Ultimately, these strategies aim to enhance patient outcomes and safety through meticulous oversight.
Related Posts
-
Why Thoracolumbar Implant Systems Are Essential for Spinal Surgery?
-

Top 10 Implant System Portfolio Choices for Dental Professionals?
-

10 Essential Tips for a Successful Minimally Invasive Spine Surgery Experience?
-

Top 10 Effective Lumbar Spine Solutions You Need to Know?
-
Top 10 Implant System Portfolios for Global Buyers in 2026?
-

Top 10 Thoracolumbar Implant Systems in China for Effective Spine Surgery?
